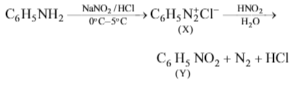Nitrogen is obtained by the thermal decomposition of :
-
Solution
Nitrogen is obtained by the thermal decomposition of
NH4NO2
NH4NO2 \(\overset{\Delta }{\rightarrow}\) N2 + 2H2O
In which of the following compound electrovalent, covalent and co-ordinate bonds are present?
-
Solution

The oxidation number of sulphur in S8, S2F2, H2S respectively, are
-
Solution
(i) Oxidation state of element in its free state is zero.
(ii) Sum of oxidation states of all atoms in compound is zero.O.N. of S in S28= 0; O.N. of S in S2F2= + 1;O.N. of S in H2S = –2;
The approximate percentage of silica in cement is:
-
Solution
The approximate composition of cement is:
Lime (CaO) = 50 – 60%
Silica (SiO2) = 20 – 25 %
Alumina (Al2O23) = 5 – 10 %
Magnesia (MgO) = 2 – 3%
Ferric oxide (Fe2O3) = 2 – 3%
An asymmetric carbon atom is that in which :
-
Solution
An asymmetric carbon atom is that in which all different groups are attached with carbon. Option (c) is correct.
Among acetic acid, phenol and n-hexanol, which of the following compounds will react with NaHCO3 solution to give sodium salt and carbon dioxide ?
-
Solution
among acetic acid, phenol and n-hexanol only CH3COOH reacts with NaHCO3 to evolve CO2 gas.
CH3COOH + NaHCO3 → CH3COONa + CO2 + H
2O
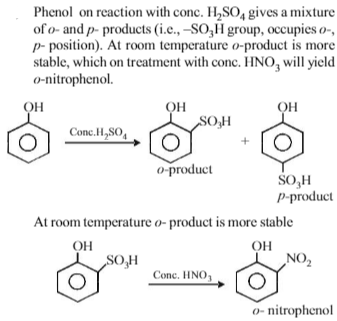
Phenol, when it first reacts with concentrated sulphuric acid and then with concentrated nitric acid, gives
-
Solution

Of the five isomeric hexanes, the isomer which can give two monochlorinated compounds is
-
Solution

Since it contains only two types ofH-atoms hence it will give only two monochlorinated compounds viz.

Be2C and Al4C3 are called –
-
Solution

A solution containing 2.675 g of CoCl3. 6 NH3(molar mass =267.5 g mol–1) is passed through acation exchanger. The chloride ions obtained in solution were treated with excess of AgNO3 to give 4.78 g of AgCl (molar mass = 143.5 g mol–1). The formula of the complex is
(At. mass of Ag = 108 u)
-
Solution

DIRECTIONS : These are Assertion-Reason type question.Question contains two statements:Statement-1 (Assertion) and Statement-2 (Reason). Answer these questions from the following four options.
Statement-1 :According to Freundlich:\(\frac{x}{m} = kp^{\frac{1}{n}}\)
Statement-2 :The isotherm shows variation of the amount of gas adsorbed by the adsorbent with temperature.
-
Solution
Statement - 1 is true but Statement - 2 is false.Freundlich adsorption isotherm gives an empirical relationship between the quantity of gas adsorbed by unit mass of solid adsorbent and pressure at a particular temperature.
Uncertainty in the position of an electron (mass = 9.1 × 10–31kg)moving with a velocity 300 ms-1, accurate upto 0.001% will be(h = 6.63 × 10-34Js)
-
Solution
Given m = 9.1 × 10-31 kg
h = 6.6 × 10-34 Js
\(\Delta v = \frac{300 \times 0.001}{100} = 0.003 \; ms^{-1}\)
From Heisenberg's uncertainity principle
\(\Delta x = \frac{6.62 \times 10^{-34}}{4 \times 3.14 \times 0.003 \times 9.1 \times 10^{-31}} = 1.92 \times 10^{-2} \; m\)
On mixing, heptane and octane form an ideal solution. At 373 K, the vapour pressures of the two liquid components(heptane and octane) are 105 kPa and 45 kPa respectively. Vapour pressure of the solution obtained by mixing 25.0 g of heptane and 35 g of octane will be (molar mass of heptane= 100 g mol-1 and of octane = 114 g mol-1)
-
Solution

In physical adsorption, gas molecules are bound on the solid surface by
-
Solution
In physisorption, gas molecules are held on the solid surface by weak van der Waal’s forces.
Which of the following halides is least stable and has doubtful existence?
-
Solution
The stability of +2 oxidation state increases and +4 oxidation state decreases as we move down in Group-14. So Pb(IV) is strongly oxidising and also I- is strong reducing agent. Hence PbI4 is non existing.
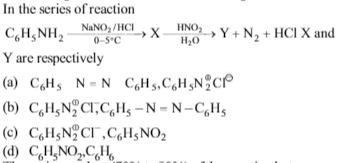
-
Solution

Which of the following compounds would be the mainproduct of an aldol condensation of acetaldehyde and acetone?
-
Solution

The rate of a chemical reaction doubles for every 10°C rise of temperature. If the temperature is raised by 50°C, the rate of the reaction increases by about :
-
Solution
Since for every 10ºC rise in temperature rate doubles for 50ºC rise in temperature increase in reaction rate= 25 = 32 times
In pyrophosphoric acid, H2P2O7, number of ? and d? – p? bonds are respectively
-
Solution

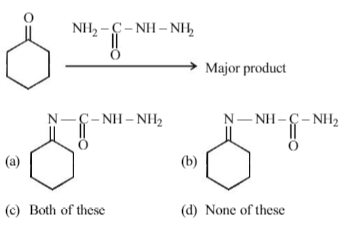
-
Solution